|
2/27/2023 0 Comments Atomic radius of calcium London: Taylor & Francis, 2002.ĭebessai, M., J. Understanding the Properties of Matter, 2nd edition. "Basal Metalliferous Sediments from the Eastern Pacific." Geological Society of America Bulletin, volume 87, Oxford: Oxford University Press, 1989.Ĭronan, D. The Elements: Their Origin, Abundance and Distribution. "Covalent Radii Revisited." Dalton Transactions, number 21, 2008, pp 2832–2838. Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán, and Santiago Alvarez. Cambridge: RSC Publishing, 2005.Ĭordero, Beatriz, Verónica Gómez, Ana E. 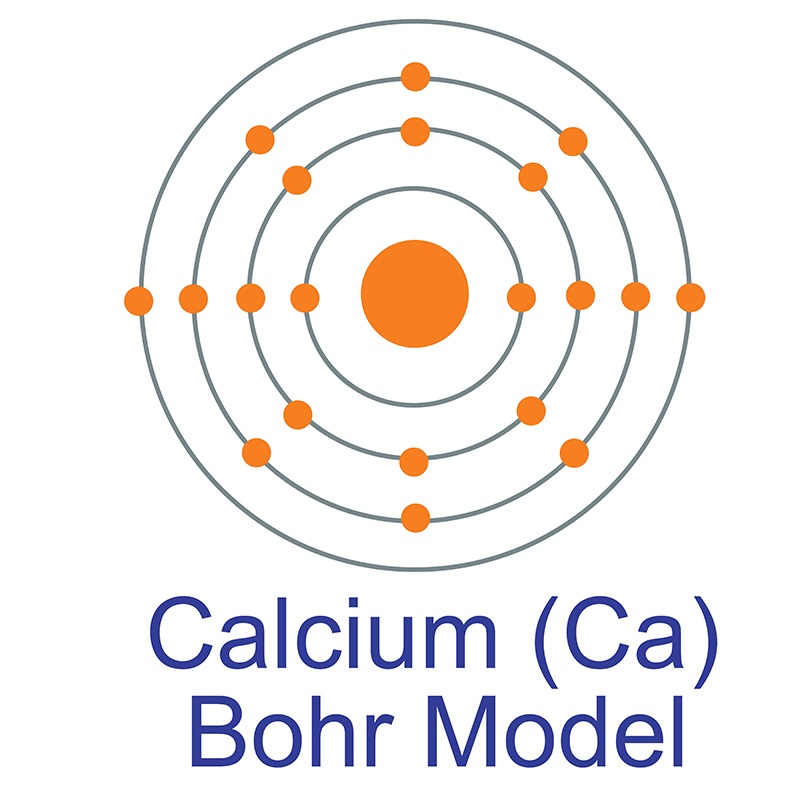
Nomenclature of Inorganic Chemistry: IUPAC Recommendations 2005. 
New York: Springer-Verlag New York, Inc., 2003.Ĭonnelly, Neil G., Ture Damhus, Richard M. "Atomic Screening Constants from SCF Functions." Journal of Chemical Physics, volume 38, number 11, 1963, pp. 2686–2689. "Electrical Resistivity of Alkaline Earth Elements." Journal of Physical and Chemical Reference Data, volume 8, number 2, 1979, pp. 439–498.Ĭlementi, E., and D. Woodbury, NY: American Chemical Society and the American Institute of Physics,Ĭhi, T. 9: NIST-JANAF Thermochemical Tables, (Part I and Part II). Berlin: Springer-Verlag, 1978.Ĭhase, Malcolm W., editor. London: Springer–Verlag, 2008.Ĭardona, M., and L. Materials Handbook: A Concise Desktop Reference, 2nd edition. Calculation and Conversion to Pauling Units." Journal of Chemical Education, volume 65, number 1, 1988, pp. 34–41. "Revised Mulliken Electronegativities: I. "Atomic and Group Electronegativities from the Electron-Density Distributions of Molecules." Journal of the American Chemical Society, volume 110, number 13, 1988, pp 4182–4186. London: Academic Press, Inc., 1979.īoyd, Russell J., and Kenneth E. "Van der Waals Radii of Elements." Inorganic Materials, volume 37, number 9, 2001, pp. 871–885. "Binding Energies in Atomic Negative Ions: III." Journal of Physical andĬhemical Reference Data, volume 28, number 6, 1999, pp. 1511–1533.īatsanov, S. "Abundances of the Elements: Meteoritic and Solar." Geochimica et Cosmochimica Acta, volume 53, number 1, 1989, pp. 197–214. 
"A Scale of Electronegativity Based on Electrostatic Force." Journal of Inorganic and Nuclear Chemistry, volume 5, number 4, 1958, pp. 264–268. "Electronegativity Values from Thermochemical Data." Journal of Inorganic and Nuclear Chemistry, volume 17, number 3-4, 1961, pp. 215–221. "Electronegativity Is the Average One-Electron Energy of the Valence-Shell Electrons in Ground-State Free Atoms." Journal of the American Chemical Society, volume 111, number 25, 1989, pp. 9003–9014. New York: Oxford University Press, 1992.Īllen, Leland C. the cationic or anionic radii.References (Click the next to a value above to see complete citation information for that entry)Īlbright, Thomas A., and Jeremy K. The atomic radii is comparatively larger than the ionic i.e. Note: The ionic radii is different from that of ionic radii, it’s often seen that we get confused between both. So here if we take the case of $C$ will be having the smaller as it possess more electron to pull inward and decreases the size. Since in a period no shell is introduced but instead electrons are filled in the same shell which increases the effective nuclear charge and the electron shielding will remain the same. if we move along a period, the atomic radii decreases along a period. 
If we move from left to right along a periodic table i.e. If we move top to bottom along a group, then we can see that the atomic radii increases from top to bottom of the group since a new shell is introduced in the elements as we move down the group. As in the question the discussion is about the atomic radii, we will discuss how atomic radii change along periods and groups of the periodic table. There are 18 groups and 7 periods in a periodic table. We know that the periodic table is the table in which the elements are placed according to the increasing order of atomic numbers. So in the question, it is asked why calcium ions possess a smaller ionic radius than the potassium ion.įor solving this question we should know the atomic numbers of the elements of the ions given.Īnd we should know some idea about the electronic configuration of the ions given here. The species of atoms, molecules or ions that possess the same valence electrons are called isoelectronic configuration. Hint: The atomic number of Ca is 20 and that for K is 19. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
